
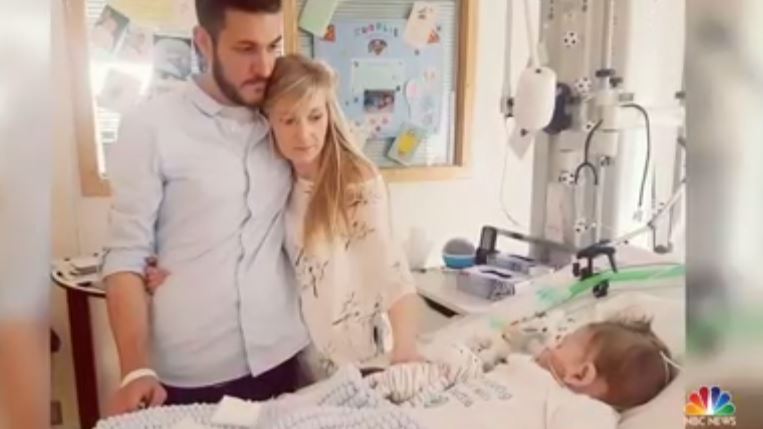
LONDON (BP) — The heart-wrenching case of a British infant with a genetic disease has triggered an international discussion about who should make end-of-life decisions for children.
“The first principle of medical ethics is ‘do no harm,'” said C. Ben Mitchell, Graves Professor of Moral Philosophy at Union University. “And this is the rub in Charlie’s case. No one wants to see him harmed, but they have disagreed at times about what’s least harmful.”
Gard’s condition is called mitochondrial DNA depletion syndrome and prevents cells from producing the energy necessary to sustain organs, according to The New York Times. He is blind and deaf, cannot breathe without a ventilator, cannot move unaided and suffers from seizures.
The infant’s parents raised $1.7 million for the experimental treatment, and an unidentified U.S. neurologist who developed the therapy has offered to administer it, The Times reported. Eighteen other patients have received the treatment.
The hospital had refused to try the therapy but said in a statement it received “fresh evidence” from “two international hospitals and their researchers” that the therapy might help.
Previously, the hospital claimed the experimental treatment likely would be futile and result in unnecessary pain, according to The Independent. Still, a Baltimore family has reported the drug helped a child with a less severe form of Gard’s condition, according to The Times.
The hospital also had refused requests by Gard’s parents to let their child die at home, LifeNews.com reported.
The refusals to grant parental requests generated protests in America and Europe.
“In cases like these the standard is ‘the best interest of the patient,'” Mitchell, a bioethicist, told Baptist Press in written comments.
“Sometimes it’s not clear what course of treatment is best or whether it’s best not to treat at all,” he noted. “”However, in consultation with his doctors, Charlie’s parents are the ones who should determine what is best for Charlie. The fact that there might be an experimental treatment that could offer some benefit to Charlie still gives the parents some hope.”
In the U.S., courts “override the parents’ wishes” for medical treatment of minors only on a “rare” basis, Mitchell said. U.K. courts are more inclined to intervene in disputes between patients and doctors, according to media reports.
A New York hospital offered July 6 to admit Gard and administer the experimental treatment. New York-Presbyterian/Columbia University Medical Center officials also said in a statement they would be willing to ship the experimental drug to London with the British hospital’s permission, The Times reported.
Both U.S. President Donald Trump and Pope Francis have weighed in on Gard’s case.
Trump tweeted July 3, “If we can help little #CharlieGard … we would be delighted to do so.” He was scheduled to speak with British Prime Minister Teresa May about the matter July 7 at the G20 Summit in Germany, according to media reports.
The pope said Gard’s parents should be allowed to “accompany and treat their child until the end,” and the Vatican’s pediatric hospital offered to admit Gard, The Telegraph reported. British officials declined the offer of hospital admittance.
Joy Riley, a physician who serves as executive director of the Tennessee Center for Bioethics & Culture, told BP in written comments, “This is a hard case brought to the public eye.”
“When a case has reached this level of struggle,” Riley said, “it is difficult as caregivers to step out of the entrenched positions to ask some very important questions, such as:
— “On whom are we focused — ourselves, or the patient and his family?
— “What are the goals of the patient and his family? (In the case of dependent minors, the parents are the usual proxy decision-makers.)
— “Is the patient being harmed by trying to meet these goals?
— “Can we find common ground?
— “If there is basic disagreement about the goals, can we refer the patient/his family elsewhere?”
















